BENZOYL PEROXIDE
PRODUCT IDENTIFICATION
CAS NO.
94-36-0
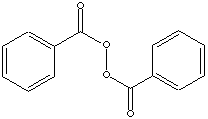
242.23
CLASSIFICATION
EXTRA NOTES
A peroxide derivative that has been used topically for BURNS and as a dermatologic agent in the treatment of acne and poison ivy dermatitis. It is used also as a bleach in the food industry.
Widely used initiator, curing agent, and cross-linking agent in polymerization processes
PHYSICAL AND CHEMICAL PROPERTIES
white powder
1.33 - 1.35
50 C
EXTERNAL LINKS & GENERAL DESCRIPTION
ftp://ftp.fao.org/
It
is used as a bleaching agent for certain foods, an oxidizing agent,
a polymerizing initiator in the manufacture of plastics, a curing
agent for silicone rubber, a constituent of ointments for skin disorders,
and an ingredient in various industrial processes. Benzoyl peroxide
has a long history of use in the food industry as a bleaching agent
added for flour, whey, and milk for cheese making. A premix of 32%
benzoyl peroxide and 68% cornstarch is used in bleaching flour.
The maximum amount used as a flour bleaching agent is 50 mg/kg.
Benzoyl peroxide has been evaluated in the 7th JECFA meeting to
an unconditional acceptance zone at 0 – 40 mg/kg and conditional
acceptance level of 40 – 75 mg/kg for treatment of flour to be consumed
by man (WHO, 1964). It has been reported that benzoyl peroxide is
typically used in the cheese manufacture at a level of 20 mg/kg
to bleach milk used for the production of white Italian cheeses
(Asiago fresh, Asiago soft cheese, Asiago medium cheese, Asiago
old cheese, Blue cheese, Caciocavallo siciliano cheese, Gorgonzola
cheese, Parmesan and Reggiano cheese, Provolone cheese, Romano cheese,
Swiss and Emmental cheese) (U.S. FDA, 2003b). The FDA has affirmed
benzoyl peroxide to be GRAS when used as a bleaching agent, following
current GMP conditions of use, for the above-mentioned foods (U.S.
FDA, 2003a).
Local: Peroxide: Compound containing the peroxy group (-O-O-), chainlike structure, containing two oxygen atoms, each of which is bonded to the other and to a radical or some element. It is considered that hydrogen peroxide is the starting material to prepare organic and inorganic peroxides commercially. Hydrogen Peroxide H2O2, is a powerful oxidizing agent. The most valuable property of hydrogen peroxide is that it breaks down into water and oxygen and therefore does not form any persistent, toxic residual compounds. It is used in the processes of epoxidation, oxidation, hydroxylation and reduction. Its oxidizing properties are used in the bleachings and deodorizing for textile, hair and in paper manufacture. It is also used medicinally as an antiseptic. Its application involves the production of chemicals like perhydrates as well as organic peroxides in which some organic (or inorganic) substituents have replaced one or both hydrogens. Some metals form peroxides in air sodium, barium or zinc. Metal peroxide releases oxygen slowly in contact with atmospheric moisture and used to as disinfectants in cosmetics, detergents, toothpaste and pharmaceuticals. They can be used in the bleachings and deodorizing and a oxygen release source in agricultural application to generate contaminated soils and lakes. Organic Peroxides are powerful oxidizing agents releasing oxygen. They are widely used as initiators,catalysts and crosslinking agent for the polymerization process in the plastics manufacturing industry and as chemical intermediates, bleaching agents, drying and cleaning agents. They are also used as antiseptics, disinfectants and germicides medically for cosmetics, detergents, toothpaste and pharmaceuticals. Organic peroxides are classified in peroxydicarbonates, peroxyketals, peroxyesters, ketone peroxides, hydroperoxides, dialkyl peroxides, diacyl peroxides by HMIS.
Benzoyl peroxide consists of two benzoyl groups linked by a peroxide chian. It is one of the general polymerization initiators for ethylene, vinylacetate, acrylates. PVC, unsaturated polyesters. It is used as a catalyst in oxidation and sulfonation reactions. It is also used in bleaching, disinfectant, drying, and cleaning agent. It is a strong free radical source containing more than 4.9% of active oxygen. The half-life temperatures are 73 C (for 10 hours), 92 C (1 for 1 hour) and 131 C (for 1 minute). It decomposes rapidly, causing fire and explosion hazard, on heating and under influence of light. It reacts violently with incompatible substances or ignition sources (acids, bases, reducing agents, and heavy metals). It should be stored in a dry and refrigerated (< 27C recommended or 38 C max) area and to keep away from reducing agents and incompatible substances.
In pharmaceutical and food field, the release of oxygen from benzoyl peroxide provide an important application to use as a topical antibacterial used for burns and as a dermatologic agent in the treatment of acne and poison ivy. It shows drying and desquamative actions that promote evacuation of comedones. It has keratolytic function that promotes keratolysis (softening and dissolution or peeling of the horny layer of the epidermis). Topical keratolytic agents are beta hydroxy acids such as salicylic acid. Benzoyl peroxide is also used as an oxidizing agent in the treatment of decubital and stasis ulcers.
KERATOLYTIC AGENTS
|
Keratolytic |
CAS RN |
| Acitretin | 55079-83-9 |
| Alcloxa | 1317-25-5 |
| Aldioxa | 5579-81-7 |
| Allantoin | 97-59-6 |
| Benzoyl peroxide | 94-36-0 |
| Dibenzothiophene | 132-65-0 |
| Etarotene | 87719-32-2 |
| Etretinate | 54350-48-0 |
| Isotretinoin | 4759-48-2 |
| Motretinide | 56281-36-8 |
| Nordihydroguaiaretic acid | 500-38-9 |
| Podofilox | 518-28-5 |
| Podophyllum resin | 9000-55-9 |
| Resorcinol | 108-46-3 |
| Resorcinol monoacetate | 102-29-4 |
| Salicylic acid | 69-72-7 |
| Sumarotene | 84264-84-6 |
| Sumarotene | 105687-93-2 |
| Tazarotene | 118292-40-3 |
| Tetroquinone | 319-89-1 |
| Tretinoin | 302-79-4 |
APPEARANCE
white granule
PEROXIDE CONTENT
75.0 - 78.0% (PEROXIDE ON DRY BASIS: 99.0% min)
WATER
21.0 - 25.0%
FREE CHLORIDE
0.1% max
FREE ACID
0.5% max
ACTIVE OXYGEN
6.62% (Theoretical)
PRICE INFORMATION